1. What Was the Chemical Effect of the Added Co2 on the Phenol Red Solution? How Do You Know?
In this experiment, students use their own exhaled jiff to explore the reaction between carbon dioxide and water. They observe the formation of a weak acid via the color change of an acrid–base indicator
This is a relatively cursory and straightforward exploration of the reaction of carbon dioxide and water at a elementary level, which should have no more than 15 minutes.
When carbon dioxide reacts with water a weak acid is formed. Carbon dioxide nowadays in exhaled air is diddled into a flask containing an indicator sensitive to small changes of pH in the appropriate region of the pH scale, and the consistent colour changes observed and recorded. The equation for the reaction between carbon dioxide and water may exist introduced for advisable students.
If students have non however met the compositions of inhaled and exhaled air, this experiment tin can serve equally function of the learning sequence for the topic of breathing and respiration in an introductory science course, using an appropriately elementary approach to the chemistry involved.
For students who have already covered the topic of breathing and respiration, and know that carbon dioxide is a significant component of exhaled air, the focus in this experiment can be transferred to the nature of the chemical reaction (other related topics could exist acrid rain, gas liquid reactions or indicators).
The equation for the reaction betwixt carbon dioxide and h2o may be introduced for advisable students.
Equipment
Appliance
- Eye protection
- Conical flask, 250 cm3, x2
- Indicator bottles with dropping pipettes, x3
Chemicals
- Ethanol (IDA – Industrial Denatured Alcohol) (HIGHLY Flammable, HARMFUL)
- Thymolphthalein indicator solution (HIGHLY Combustible), access to modest bottle with dropper
- Phenol ruddy indicator solution (HIGHLY Combustible), access to small canteen with dropper
- Sodium hydroxide solution, 0.4 M (IRRITANT), small bottle with dropper
- Distilled (or deionised) h2o, 125 cm3, x2
Health, safety and technical notes
- Read our standard health and safety guidance.
- Article of clothing eye protection throughout.
- Phenol red indicator – see CLEAPSS Hazcard HC032. The indicator may be purchased every bit a solid reagent or as a set-made solution in ethanol. The solution may exist made from the solid reagents by preparing a 5% w/5 solution in ethanol (IDA). If 30 cm3 or 60 cm3 dropping bottles with integral dropping pipettes are available, these are platonic for dispensing the indicator solutions. While phenol red itself is not flammable, its solution in ethanol is highly flammable.
- Thymolphthalein indicator – run across CLEAPSS Hazcard HC032. The indicator may exist purchased as a solid reagent or equally a fix-made solution in ethanol. The solution may be fabricated from the solid reagents past preparing a 5% due west/v solution in ethanol (IDA). If 30 cm3 or sixty cmiii dropping bottles with integral dropping pipettes are available, these are ideal for dispensing the indicator solutions. While thymolphthalein itself is not combustible, its solution in ethanol is highly flammable.
- Ethanol (IDA – Industrial Denatured Alcohol), CHiiiCH2OH(l), (HIGHLY FLAMMABLE, HARMFUL) – see CLEAPSS Hazcard HC040A.
- Sodium hydroxide solution, NaOH(aq), (IRRITANT at concentration used) – meet CLEAPSS Hazcard HC091a and CLEAPSS Recipe Book RB085.
Process
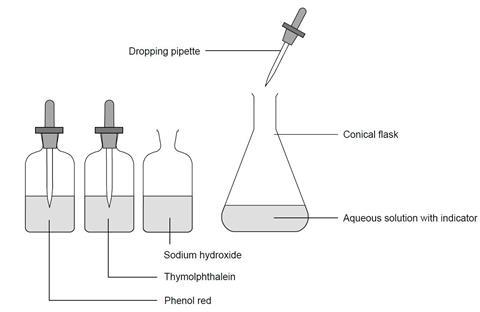
Activity 1
- Identify about 125 cm3 of h2o in a 250 cmthree conical flask.
- Add five or half dozen drops of thymolphthalein indicator to the water.
- Add together but enough sodium hydroxide solution (about two or 3 drops) to produce a blue colour.
- Talk or blow gently into the flask – ie add the carbon dioxide.
- Go along calculation the carbon dioxide until a colour change is observed.
Activity 2
- Place about 125 cm3 of h2o in a 250 cm3 conical flask.
- Add i or two drops of phenol red to the water.
- Add 2 drops of sodium hydroxide solution to produce a carmine solution.
- Talk or blow gently into the flask – ie add together carbon dioxide.
- Continue adding the carbon dioxide until a colour modify is observed.
Questions for the form
- Why does the color change not occur instantly?
- What is the reason for adding a few drops of sodium hydroxide solution (NaOH) earlier each experiment?
Answers to questions
- The amount of carbon dioxide in each breath is small, so it takes a lot of breaths to react with the alkali.
- To ensure the solution is slightly alkaline metal at the beginning and to neutralise any COtwo or any other acid initially nowadays.
Instruction notes
Straws are non necessary for bravado exhaled air into the flask; simply breathing or speaking into the flask is sufficient to cause the indicator to change colour.
Phenol red indicator changes from yellow to cherry-red over the pH range 6.viii–8.4. Thymolphthalein (the alternative bromothymol blue could too be used) changes from bluish (alkaline) to colourless (acid) over the pH range nine.3–10.5. Come across CLEAPSS Recipe Book RB000, which also covers bicarbonate indicator solution.
Eventually sufficient carbon dioxide from the students' breath dissolves and produces enough acid in the solution to change the colour of the indicator:
CO2(aq) + HtwoO(l) ⇌ H+(aq) + HCO3 –(aq)
CO2 too reacts with NaOH. This reaction produces the less alkaline Na2CO3:
2NaOH(aq) + CO2(g) → NaiiCO3(aq) + H2O(l)
The equilibrium betwixt carbon dioxide and water can be reversed by heating the weakly acidic solution to just below boiling. The solubility of carbon dioxide in h2o decreases as the temperature is raised, and it is driven off into the atmosphere. The concentration of dissolved carbon dioxide therefore drops, causing the equilibrium to shift to the left and the indicator colour to change back to carmine. On cooling the solution and blowing exhaled breath into the flask again, the sequence can be repeated.
hornpingerced1936.blogspot.com
Source: https://edu.rsc.org/experiments/the-reaction-of-carbon-dioxide-with-water/414.article
0 Response to "1. What Was the Chemical Effect of the Added Co2 on the Phenol Red Solution? How Do You Know?"
Post a Comment